AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Plum pudding model2/13/2024 
Thomson's proposal, based on Kelvin's model of a positive volume charge, served to guide future experiments. Thomson based his atomic model on known experimental evidence of the day, and in fact, followed Lord Kelvin's lead again as Kelvin had proposed a positive sphere atom a year earlier. Thomson abandoned his 1890 "nebular atom" hypothesis, based on the vortex theory of the atom, in which atoms were composed of immaterial vortices and suggested there were similarities between the arrangement of vortices and periodic regularity found among the chemical elements. He had followed the work of William Thomson who had written a paper proposing a vortex atom in 1867, J.J. Thomson's model was the first to assign a specific inner structure to an atom, though his original description did not include mathematical formulas. the atoms of the elements consist of a number of negatively electrified corpuscles enclosed in a sphere of uniform positive electrification. Thomson published his proposed model in the March 1904 edition of the Philosophical Magazine, the leading British science journal of the day. 
Thomson held that atoms must also contain some positive charge that cancels out the negative charge of their electrons. It had also been known for many years that atoms have no net electric charge. Stoney had coined for the " fundamental unit quantity of electricity" in 1891. Thomson called them "corpuscles" ( particles), but they were more commonly called "electrons", the name G. It had been known for many years that atoms contain negatively charged subatomic particles. The plum pudding model has electrons surrounded by a volume of positive charge, like negatively charged "plums" embedded in a positively charged " pudding". Thomson in 1904 soon after the discovery of the electron, but before the discovery of the atomic nucleus, the model tried to account for two properties of atoms then known: that electrons are negatively charged subatomic particles and that atoms have no net electric charge. The plum pudding model is one of several historical scientific models of the atom. 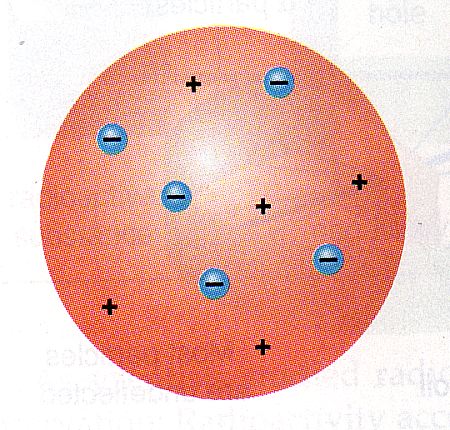
Obsolete model of the atom The plum pudding model of the atom The current model of the sub-atomic structure involves a dense nucleus surrounded by a probabilistic "cloud" of electrons
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
